 Still working on chemistry this week. Our warm up today is a review of drawing atoms and identifying the number of protons, electrons, and neutrons. Thursday and Friday we will have a formal quiz that covers the atom. You must have your periodic table that day to use for the quiz. Next, we did a round robin around the room using the Atomic Structure Task Cards as a review of atomic structures. This assignment was on page 13 in your notebook. If you missed today, please make time during homeroom to come to my room to complete this assignment or here is a powerpoint of the assignment.
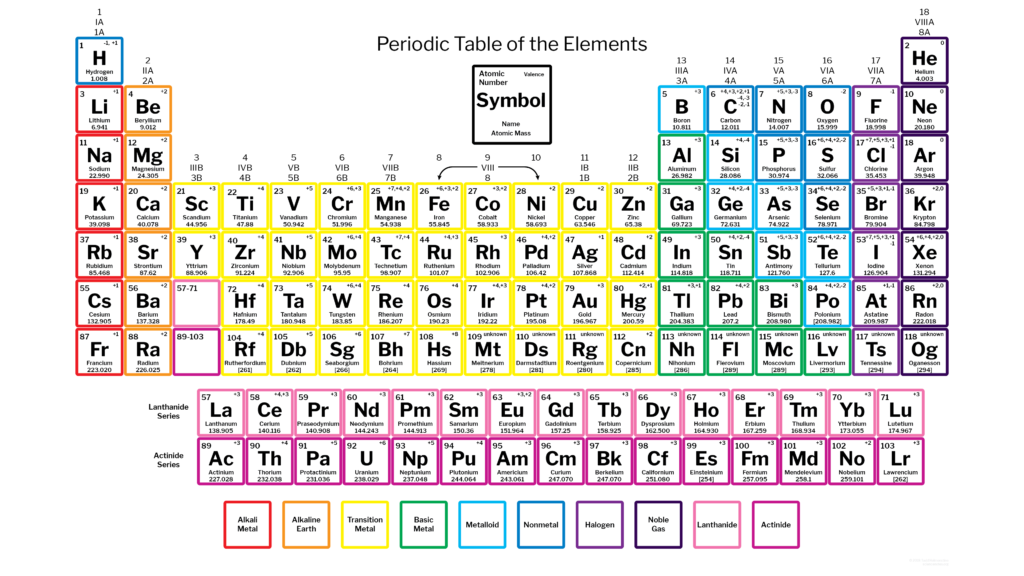
Lastly, we had a list of things to accomplish today: 1. Schoolnet practice quizzes(open notes) on ATOMS and ATOMIC MASS - these will go down as informal grades. The schoolnet codes will be given during class. 2. We read a passage on metals, non metals and metalloids. Students created vocabulary pyramids (on page 14) for each of the vocabulary words and colored in a blank periodic table according to metals, non metals and metalloids. Click here for the reading passage for today. - Read the passage - you do not need to annotate - Complete the vocabulary pyramids 3. Color in the periodic table 3 colors and make a key that indicates the metals, non metals and metalloids. You can either use the reading passage to color the periodic table or you can use page D20 in the textbook. Attach the periodic table to the bottom of the back of page 14. IF YOU DID NOT FINISH STEPS 1-3...FINISH THEM FOR HOMEWORK  QUIZ THURSDAY AND FRIDAY on ATOMS!! MAKE SURE YOU BRING YOUR PERIODIC TABLE If you are unhappy with your annotation grade on the Mendeleev assignment, snap shot the first two paragraphs of the following sections and resubmit them, You should have 3 files attached. If your goal is to improve your grade it needs to be annotated THOROUGHLY, not just one comment per paragraph.
Put in order of Increasing Atomic Number Building on Others' Acheivements Completeing the Puzzle Next, we continued our notes on page 11 on ATOM DRAWINGS. If you were not here today, please get the notes in your notebook and come see me during homeroom if you need extra help or ask a peer for help. If you were not here for the notes, you can find a copy here.
After we practiced drawing atoms, each student created a model of one of the atoms on the periodic table. After completing the model, students chose 8 atoms from the handout Reading the Periodic Table on page 12 in our notebooks and practiced drawing those atoms on a separate sheet of paper to be turned in next class. If you lost your copy, here is the handout. Finish drawing the atoms for homework!!!
Next, we will be starting our first unit on Chemistry. We set up our notebooks for Chemistry. Title: CHEMISTRY ...8 Table of contents....9 Introduction to the Atom...10 We introduced Chemistry with an article on Mendeleev, the Russian scientist who gets credit for creating the idea of the Periodic table. We will be reading this article on Newsela.com. This is a site that has articles on all kinds of topics in science, math and social studies. For our class, you will use class code: P8DS9B Read and annotate the passage on Mendeleev. Answer the following questions on the back of page 10 in your notebook. Do not complete the quiz at the end of the reading. 1.What was so amazing about Mendeleev’s first periodic table? 2.What is the difference between organic and inorganic chemistry? 3.Originally, how did Mendeleev organize his periodic table? 4.Why did Mendeleev call the table of elements, “periodic”? 5.Ultimately, how did Mendeleev end up organizing the periodic table? 6.Why did Mendeleev get all the credit for the periodic table? 7. Why are the lanthanides and the actinides at the bottom of the periodic table? 8.What is the difference between periods and groups?
Students worked on the handout Reading the Periodic Table on the back of page 10 in our notebooks. If you lost your copy, here is the handout. Finish this assignment for homework!!!
Our warm up today is a review of scientific method. Next, we practiced writing the parts of the scientific method using magazine advertisements. Students were expected to look at an ad, and design an experiment based on what the ad was trying to tell us. They needed to include the question they were trying to answer, the procedure, the independent variable, the dependent variable, control group, experimental group, and constants. We created posters of the "planned"experiment.
Next we will be starting our first unit on Chemistry. We set up our notebooks for the unit. The beginning of each unit will be marked by a new cover sheet and a new table of contents. Title: CHEMISTRY ...8 Table of contents....9 Introduction to the Atom...10 HOMEWORK: Lastly, the students were given the Bart Simpson Handout to practice the Scientific Method. If you lost your copy...here is the handout you can print out. It is due next class. Fourth block B day---If you did not finish in class, here are the questions. Please turn it in Friday during class.
One last mini lab today using the balances and also as an intro to the scientific method. We tried to identify how much sugar is found in bubble gum! We were surprised to see how high the % of sugar turned out to be in the gum. Next we took notes on page 7 in our notebooks on Scientific Method. If you were absent, here are the notes from today's powerpoint. In small groups we went through an experiment and identified the steps of the scientific method. Next we did a practice problem as a group on scientific method, and turned in one copy for a group grade. Next, we practiced writing the parts of the scientific method using magazine advertisements. Students were expected to look at an ad, and design an experiment based on what the ad was trying to tell us. They needed to include the question they were trying to answer, the procedure, the independent variable, the dependent variable, control group, experimental group, and constants. We created posters of the "planned"experiment.
A great way to the end the week. Our warm up today was a review of qualitative and quantitative observations. Reminder, Qualitative observation is a subjective gathering of information which focuses more on differences in quality rather than differences in quantity. Quantitative observation, on the other hand, is an objective gathering of information which focuses on numbers or measurements while basing results on statistics and numeric analyses. Next, we completed the lab on crystals that we started last class. Last class we made observations on the crystals and this class we slowly added 10 mL of water at a time. While adding water to the crystals we continued to add observations to our notebooks. We tried to add both qualitative and quantitative observations. Next we tried to infer the function of crystals and the purpose they are sold. Next, we took a quick mini-quiz on mass and volume(informal grade). Grades should be in powerschools this afternoon.
Lastly, we started taking notes on the Scientific Method on page 7 in our notebooks. If you were not here last class, here is the powerpoint on Scientific Method that we started. We will finish the notes on Tuesday and start a lab to practice the scientific method. Our warm up was a review of reading graduated cylinders and balances. We will have a very short quiz next class to make sure everyone can calculate mass and volume. Next we did one more practice with balancing a set of cubes that all have the same volume but different masses.
We also organized our notebooks today. If you were not here or did not have your notebook, here is the list of page numbers and what handouts should go on which page. •Tape the green brochure on the back of page 1 •Tape your textbook scavenger hunt on the front of page 2 •All About Me A-Z…back of page 2 •Tape Mass notes to the front of page 3 •Tape Volume Notes on page 4 •Tape Color Challenge to the back of page 4 Next, we took notes on observations and inferences on page 5 in our notebooks. Click here are the notes that should go on page 5. Finally we did a lab using unknown crystals. The purpose of this lab was to practice making qualitative and quantitative observations. Reminder : Quiz Friday/Monday on Mass and Volume - Reading the balances and the grad. cylinders -Know the units -Know the instruments -Know the definitions |
Details
Archives
October 2018
Categories |
||||||||||||||
Photo used under Creative Commons from Free Public Domain Illustrations by rawpixel





























 RSS Feed
RSS Feed
